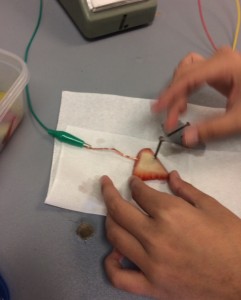
Hypothesis: When we test out the fruits; grapes, kiwi, strawberry and apple the most acidic fruit will produce the most voltage because acids make ions/charged particles, these charged particles are the same as the ones that are produced in an electric current therefore I believe that the Kiwi will produce the most volage because a kiwi is sour therefore it is acidic and makes ions. There is also a chance that the strawberries can produce the most voltage because they are acidic as well.
Experiment
Results
Strawberries: -1 (v)
Kiwi: +o.5 (v)
Apple: +0.25 (v)
Grape: + 0.25 (v)
Most Important Findings: Strawberries were the only negative number on the dial and made the most movement. Kiwi had the highest positive movement with +0.5. The negative and positives have to do with the alligator clips. If you switch them they will be opposite.
Questions
Our group did the experiment with 4 different types of fruits, kiwi, strawberry, apple and grapes. I brought in the kiwis and the strawberries. None of these fruits were able to make the bulb light up, this is because fruits don’t produce enough voltage to light up a bulb. We were able to tell how much energy it was producing through the amount of movement the arrow on the voltage meter was producing. One way to improve this experiment so that the results could be more accurate would be to use the same size of piece of fruit for each type because maybe if we had a bigger sized kiwi it would have been able to light up the bulb, but the apple was way bigger than the grape and maybe that’s why the apple did better. We could also be more accurate with the angle we put the nail into the fruit.
Electricity is produced by a chain of electrons moving from point a to b to create a current. In this experiment electricity was produced from the acid in the fruits. The type of reaction that occurs in fruits so that they are able to create electricity is oxidation-reduction. There are two types of cells that can facilitate this type of reaction, galvanic cells and electrolyte cells. Galvanic cells are spontaneous cells that act as the battery and are able to create a charge by themselves. Electrolyte cells require electricity in order for them to have a charge. This is experiment used a closed circuit because it consisted of a series of wires connecting to one another and the fruit allowing electricity to flow through.
We can use this information in our everyday lives to help us realize that electricity can come from the most commonly used things in our life.
Extra observations:
- Grapes and Apples were the exact same
- We had to play around with the way we clipped the wire onto the nail so that it could power the dial
- We didn’t have time to try the lemon ourselves but it seemed that the lemon worked the best.
- We were able to do this experiment by creating a circuit
- If we had put two nails into the fruit it wouldn’t have worked because they are the same materials and would repel.